For the one poster that asked, Tecnical grade KOH can be found at Hvchemical.com
Tuesday, August 12, 2008
Particulate Comparison Between Potassium Hydroxide and Borax
I perform a comparison between potassium hydroxide and borax. I was a bit disappointed with the precipitation of the KOH before the test started. This leads me to believe my supply of KOH is not technical grade.
For the one poster that asked, Tecnical grade KOH can be found at Hvchemical.com
For the one poster that asked, Tecnical grade KOH can be found at Hvchemical.com
Monday, August 11, 2008
Operational Test of an Adjustable Pressure Relief Valve
I conduct a flashback test of a Smack's Booster style container with an adjustable pressure relief valve mounted to it to determine if the valve can control the pressures generated by a flashback.
The result is surprisingly good, considering I was afraid I'd witness a container rupture. Big bangs be bad in my book, but no big bad bangs were to be had. Boo Hoo!
The result is surprisingly good, considering I was afraid I'd witness a container rupture. Big bangs be bad in my book, but no big bad bangs were to be had. Boo Hoo!
First Use of the Rotameter
I used the rotameter for the first time and figured out that I was on the high side with the flowtube size. This means I have to order another flowtube. Darnit!
Using a Drill Press to Cut Plate Holes
I go through cutting plate holes using a drill press and ramble on about the do's, don'ts and particulars of using a drill press.
1. When running a drill press keep your hands out of the work.
2. Don't pick metal shavings out of the work with your hands while the drill is running.
3. When cutting stainless steel run the drill press at the lowest speed setting. 150-200RPM is best, or the slowest the machine will adjust to if this speed can't be obtained.
4. If the work is smoking, resort to using cutting oil.
5. Support the work with a vise. The vise HAS to be rigidly mounted to the table. Most vises have gaps in the bed so cut a piece of wood to support the work completely.
6. Make sure your drill press can do the work. A 1/3Hp 1/2" chuck press will do the job.
7. To cut Stainless Steel, use at least a Cobalt drill bit. They can be sharpened when dull where the titanium washed ones are useless once dull.
8. A used 12", 1/2" chuck bench model drill press can be surprisingly affordable if you go out looking for one. I recommend older ones made in the good old USA.
9. If buying a used drill press, check the side play of the chuck by wiggling it back and forth. If the movement is visible and clicking can be felt, then the press is worn and should be avoided.
1. When running a drill press keep your hands out of the work.
2. Don't pick metal shavings out of the work with your hands while the drill is running.
3. When cutting stainless steel run the drill press at the lowest speed setting. 150-200RPM is best, or the slowest the machine will adjust to if this speed can't be obtained.
4. If the work is smoking, resort to using cutting oil.
5. Support the work with a vise. The vise HAS to be rigidly mounted to the table. Most vises have gaps in the bed so cut a piece of wood to support the work completely.
6. Make sure your drill press can do the work. A 1/3Hp 1/2" chuck press will do the job.
7. To cut Stainless Steel, use at least a Cobalt drill bit. They can be sharpened when dull where the titanium washed ones are useless once dull.
8. A used 12", 1/2" chuck bench model drill press can be surprisingly affordable if you go out looking for one. I recommend older ones made in the good old USA.
9. If buying a used drill press, check the side play of the chuck by wiggling it back and forth. If the movement is visible and clicking can be felt, then the press is worn and should be avoided.
Plate Conditioning with a +10n- Plate Brick
I conducted plate conditioning with a +10n- plate brick. I was after conditioned plates, while at the same time I wanted to see what would happen with this particular setup. I estimated that I would have 12/11= 1.09 volts across the plate gaps. Unfortunately I must have placed the plates too close together.
Otherwise the brown particulate occurred as normal.
Otherwise the brown particulate occurred as normal.
Labels:
alternative,
brown's,
Browns,
Conditioning,
configuration,
electrolysis,
fuel,
gas,
generation,
HHO,
hydroxy
Sunday, August 10, 2008
Observational Electrolyte Test Series Summary
The Observational Electrolyte Test Series was intended to provide a body of work where the electrolytes were subjected to cooking, mostly by way of just two plates. The results were then observed and recorded. The primary intent was to observe particulate and scum formation along with additional data as it was observed. While I attempted an observational assessment of gas production, it was not a parameter or goal of the test.
To date I've tested the following electrolytes.
I note that for the purposes of this test, there were three classifications of effect and two that I recorded. Particulate is the formation of solids in the electrolyte, usually brown. Particulate usually settles on the bottom after testing with the exception of vinegar. Scum is the formation of a surface viscus mass that interferes with bubbles bursting. Scum does not seem to have a color of it's own and usually dissolves back into the water within a few minutes of electrolysis. Foam is similar to scum in the interference of bubble bursting, but without direct observation of a substance on the surface. Foam usually lingers on the surface for awhile after electrolysis. Foam measurements were not taken, but foam formation did cause me to abort the CLR test.
I determined early on that using distilled water or reverse osmosis purified water has by far the least effect on particulate formation. This in turn causes me to assert that ordinary tap water with high mineral content should be avoided when performing electrolysis. Softened water is also better than plain tap water, but is still second to distilled or reverse osmosis purified water.
Despite some inconsistencies in the tests these are the results. I may add tests to this series but felt there was enough of a body of work to produce a summary.
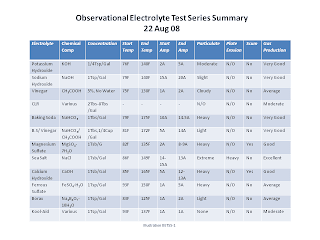
I also note that compounds containing Magnesium or Calcium should be avoided as they leave hard water deposits on the container and plates.
NOTE: Wear hearing protection when igniting a foam buildup!!! Igniting foam makes a sound at least as loud as a high caliber rifle or shotgun.
To date I've tested the following electrolytes.
- Potassium Hydroxide (KOH)
- Sodium Hydroxide (NaOH)
- Vinegar(CH3COOH)
- CLR (Calcium, Lime & Rust Cleaner, proprietary mix)
- Baking Soda (NaHCO3)
- Baking Soda/Vinegar (NaHCO3/CH3COOH)
- Magnesium Sulfate (MgSO4·7H2O)
- Sea Salt/Sodium Silicate (NaCl)
- Calcium Hydroxide (CaOH)
- Ferrous Sulfate (FeSO4·H2O)
- Borax (Na2B4O7·10H2O)
- Kool-Aid (Citric Acid, Ascorbic Acid, others)
I note that for the purposes of this test, there were three classifications of effect and two that I recorded. Particulate is the formation of solids in the electrolyte, usually brown. Particulate usually settles on the bottom after testing with the exception of vinegar. Scum is the formation of a surface viscus mass that interferes with bubbles bursting. Scum does not seem to have a color of it's own and usually dissolves back into the water within a few minutes of electrolysis. Foam is similar to scum in the interference of bubble bursting, but without direct observation of a substance on the surface. Foam usually lingers on the surface for awhile after electrolysis. Foam measurements were not taken, but foam formation did cause me to abort the CLR test.
I determined early on that using distilled water or reverse osmosis purified water has by far the least effect on particulate formation. This in turn causes me to assert that ordinary tap water with high mineral content should be avoided when performing electrolysis. Softened water is also better than plain tap water, but is still second to distilled or reverse osmosis purified water.
Despite some inconsistencies in the tests these are the results. I may add tests to this series but felt there was enough of a body of work to produce a summary.

I also note that compounds containing Magnesium or Calcium should be avoided as they leave hard water deposits on the container and plates.
NOTE: Wear hearing protection when igniting a foam buildup!!! Igniting foam makes a sound at least as loud as a high caliber rifle or shotgun.
Labels:
alternative,
brown's,
Browns,
electrolysis,
Electrolyte,
free,
fuel,
gas,
generation,
HHO,
hydrogen,
hydroxy,
particulate,
scum
Observational Electrolysis Test using Potassium Hydroxide
I conduct an observational electrolysis test using potassium hydroxide (KOH) as the electrolyte and observe the results.
My Observations
1 The electrolyte particulates out about 3/16Th of an inch before the test begins.
2. Brown particulate forms about 1/4 inch during the test.
3. Foam forms on the surface and stays there during the test.
4. Gas production is quite good.
5. Some brown particulate makes its way into the foam. It doesn't appear to be of the scum variety.
6. I used quite a bit less KOH in this test than the electrolytes in other tests.
7. I suspect my batch of KOH is an inferior grade.
Next:
Previous: Grape Kool-Aid Long Term Particulate Comparison Test
My Observations
1 The electrolyte particulates out about 3/16Th of an inch before the test begins.
2. Brown particulate forms about 1/4 inch during the test.
3. Foam forms on the surface and stays there during the test.
4. Gas production is quite good.
5. Some brown particulate makes its way into the foam. It doesn't appear to be of the scum variety.
6. I used quite a bit less KOH in this test than the electrolytes in other tests.
7. I suspect my batch of KOH is an inferior grade.
Electrolytes & Electrolyte Testing Articles
Next:
Previous: Grape Kool-Aid Long Term Particulate Comparison Test
Labels:
alternative,
brown's,
Browns,
electrolysis,
Electrolyte,
free,
fuel,
gas,
generation,
HHO,
hydrogen,
hydroxy,
particulate,
scum,
Test
Subscribe to:
Posts (Atom)


